From the Director, Prof Chris Rowe
Welcome to the August 2021 edition of our ADNeT Newsletter. Highlights in this issue include:
- FDA Approval of Aducanumab
- Australian Dementia Network Registry welcomes additional consumer representatives to its Steering Committee and Survey Working Group
- A contribution to the Australian Journal of Dementia Care, by Prof. Sharon Naismith and Prof. Graeme Samuel
- Events and webinars
- And much more.
I hope you enjoy our newsletter. You can read the newsletter here or download it from our website at https://www.australiandementianetwork.org.au/2021/08/17/adnet-news-august-2021/
If you have any stories you would like us to cover, please email adnet-info@unimelb.edu.au We look forward to hearing from you.
I hope you take the time to read our newsletter.
Chris
NEWS:
‘Controversy and Progress in Alzheimer’s Disease — FDA Approval of Aducanumab’
An interesting perspective on the recently announced approval of Aducanumab by the American Federal Drug Administration.
The article sets out conflicting perspectives on the recently approved dementia drug Aducanumab. Many welcomed the approval of aducanumab as the first drug to effectively clear amyloid beta brain plaques in people with dementia. Others have expressed concerns about the drug’s efficacy in improving cognitive functions as well as its side effects and cost (US$56,000 per year or A$76,000).
The approval of the drug followed an equally controversial path, with the FDA’s Drug Advisory Committee recommending against approval. Despite this, the FDA approved the drug under its accelerated pathway in which “evidence of biomarker changes are deemed to reasonably predict clinical benefit”.
The article also sets out the minimum conditions for the prescription of Aducanumab in the USA, including its use only in patients with mild cognitive impairment or mild dementia, positive PET scan or CSF assay, and protocols for monitoring and managing side effects.
To read the full article, you can subscribe for free.
ADNeT’s Prof. Sharon Naismith and Prof. Graeme Samuel contribute to the Australian Journal of Dementia Care


Prof. Sharon Naismith, Lead, Australian Dementia Network Memory Clinics Initiative and Professor Graeme Samuel, Chair of ADNeT Limited, were approached by the Australian Journal of Dementia Care to draw upon their knowledge and experience to answer several questions posed on the topic of: Timely diagnosis of dementia: is it happening in Australia?
Professor Naismith outlines how, “In Australia only 23.8% of public memory clinics can provide an assessment within two months. The average wait time is almost 12 weeks, compared to, for example, 35 days, 25 days and 33 days in Ireland, Canada and the United Kingdom respectively.”
Professor Graeme Samuel highlighted that, “Timely diagnosis of dementia is essential to improve care and outcomes for people living with dementia, and to make life better for their carers. Yet the average time between first symptoms and diagnosis is just over three years.”
They were joined by Dr Jane Thompson, a long-time advocate for dementia research, and Dr Meredith Gresham, who contributed their own expertise responses on how to bridge the gap between health care systems and timely diagnosis.
You can read all the articles here. And think about how you might answer these questions from Article 1 of the Journal Club Discussion Guide: https://dta.com.au/app/uploads/downloads/JC_Booklet_E11_v1_easyread.pdf
Updates:
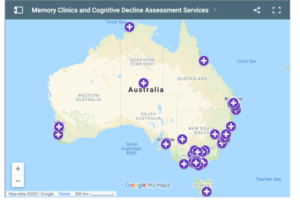
Memory Clinic Map update
126 memory clinics and specialised assessment services across the country have joined ADNeT’s list of services and can be found at www.bit.ly/adnetmap
ADNeT’s memory clinics initiative has also started to map services that offer post-diagnostic support to people with dementia and MCI (e.g., Outreach services, exercise programs, Carer support, Support Groups, etc.).
If you would like your service or clinic to be listed please contact: adnet.mc@unsw.edu.au or via https://redcap.link/q9oua992
For more information, visit https://www.australiandementianetwork.org.au/initiatives/memory-clinics-network/
Dementia Advocates shape Clinical Quality Registry operations
The Australian Dementia Network Registry (clinical quality registry) acknowledges the contribution that Dementia Australia and Dementia Advocates make to our work, bringing first-hand experience to help improve clinical care and patient outcomes for people with dementia and mild cognitive impairment. Dementia Advocates are people living with dementia and mild cognitive impairment and their carers.
Recently, Dementia Advocates have reviewed the format and content of registry participation documents. These are documents that recently diagnosed patients or their care partner receive explaining participation in the Registry. Their contribution has made the documents easier to understand while encouraging Registry participation. The updated documents have received ethics approval and will be implemented later this year following governance approval by participating sites.
We have also welcomed two new Dementia Advocates to our Steering Committee and another two to our Survey Working Group. They join existing Dementia Advocates and other members of these two important committees.
The Australian Dementia Network Registry is committed to ongoing consumer engagement and has been working with Dementia Australia to ensure that the registry incorporates the voice of people with lived experience of dementia and mild cognitive impairment.

L–R: Scott Cooper (Dementia Advocate), Sally Lambourne (General Manager, Consumer Engagement, Dementia Australia), Xiaoping Lin (Research Fellow, ADNeT Registry), Jenny Fitzpatrick (Dementia Advocate), Stephanie Ward (Geriatrician & ADNeT Registry Clinical Lead & Steering Committee Co-Chair) (Chair), Barbara Kain (Carer Representative), Yun-Hee Jeon (Susan and Isaac Wakil Professor of Healthy Ageing, University of Sydney), Kasey Wallis (ADNeT Registry Program Manager), Lee-Fay Low (Professor in Ageing and Health, University of Sydney), Henry Brodaty (Scientia Professor of Ageing and Mental Health, University of New South Wales & ADNeT Registry Steering Committee Co-Chair), Kerrie McAloney (ADNeT Registry State Coordinator).
Australian Dementia Network Registry expanding rapidly in Queensland
The ADNeT Registry network has seen a rapid expansion in Queensland with seven clinics joining the Registry in the past few months. These clinics extend the Registry’s coverage in the state from Cairns in Far North Queensland to Brisbane in the Southeast and include:
- Cairns Memory Clinic (Cairns Hospital)
- Innisfail Memory Clinic (Innisfail Hospital)
- Mareeba Memory Clinic (Mareeba Hospital)
- Memory Clinic Princess Alexandra Hospital (Princess Alexandra Hospital)
- The Prince Charles Hospital Memory Clinic (The Prince Charles Hospital)
- Neurosciences Queensland
- Robert Adam Neurology
Volunteers are the difference
ADNeT Screening and Trials initiative wants to thank the many volunteers who have signed up to participate in dementia research and trials. A large number of volunteers have already been assessed by phone, with a pass rate of 68% and 94% after the first in-person assessment. The importance of this cannot be emphasised enough. Difficulty in recruiting suitable participants in sufficient numbers means that a trial may fail to get up or that it is not available for Australian patients.
Having a large number of pre-screened, trial-ready volunteers ensures that trials can get up and running, supporting the development of treatments that are urgently needed to tackle the dementia epidemic.
Click here to find out more about how to volunteer for dementia clinical trials.
Upcoming events and webinars:
CHeBA Visiting Lecture Series | Professor Gill Livingston, University College London | All Welcome

UNSW Sydney’s Centre for Healthy Brain Ageing (CHeBA) invites you to register for the upcoming Visiting Lecture Series event:
When: Wednesday, 25 August, 2021
Time: 5:00PM to 6:00PM (AEST)
Talk Title: Preventing Dementia: What Should We Do?
Where: Zoom Webinar
The session will be followed by a live Q&A session with Professor Livingston.
For more information or assistance with registration please contact Laurie Mock
More information on past and upcoming Visiting Lecture Series talks is available here: www.bit.ly/CHeBAVisitingLectureSeries
ADNeT Webinar – Sleep disturbances and dementia
When: 26th August 2021
Time: 4-5pm
Who: Presentations from Prof. Sharon Naismith and Prof. Simon Lewis on the impacts of sleep disturbance and how it links to neurodegenerative disease.

Registrations are now open.
The inaugural Mild Cognitive Dementia (MCI) Masterclass: Dementia Australia

This Masterclass will give you an opportunity to hear the latest on MCI from world leading experts and allow you to actively contribute to the development of a best practice documents on the management of MCI.
When: Wednesday 1st September 2021.
This 1-day conference is back and open for registration after being postponed in 2020.
Where: Virtual presentation
Who: The program and speaker list is extensive and can be viewed here. ADNeT’s Prof. Chris Rowe, Prof. Sharon Naismith, Prof. Sue Kurrle, Prof. Colin Masters, and Prof. Kaarin Anstey will be presenting several sessions. Of particular note, Professor Philip Sheltens, who was recently appointed to the position of ‘Chair of the International World Dementia Council’, will be presenting on the latest breakthrough evidence for treatment of MCI.
Registration is now open to all healthcare professionals.
Alzheimer’s beyond COVID-19 – Dementia Trials Australia Annual Scientific Meeting

Dementia Trials Australia is holding a virtual Annual Scientific Meeting.
When: Friday 8 October 2021
Where: University of Sydney and online
Time: 9.15 am to 4.45pm AEST
Who: over 100 attendees comprising Principal Investigators, Sub Investigators and senior clinicians involved in Dementia Trials in Australia, including ADNeT Director, Prof. Chris Rowe and Prof. Sue Kurrle
Registration Fee: $185
View the Conference Program and get in touch with the event organiser to register your interest.
Dementia Australia’s Pedal the Map

This November Dementia Australia is organising a ‘pedal the map’ event. You can contribute your kms to travel a lap around Australia. For more information and to sign up go to: https://www.dementia.org.au/events/pedal-the-map